|
|
Nitrogen processes and cycleNitrogen is essential to life on Earth. It is a component of all proteins, and it can be found in all living systems. Nitrogen compounds are present in organic materials, foods, fertilisers, explosives, some wastewaters, and poisons. Nitrogen is crucial to life, but in excess it can also be harmful to the environment. While the nitrogen information below applies to the whole landscape, these web pages have a focus on the inputs, processes, stocks, outputs and management issues associated with aquatic/wetland systems. Quick facts
Nitrogen processes and cycle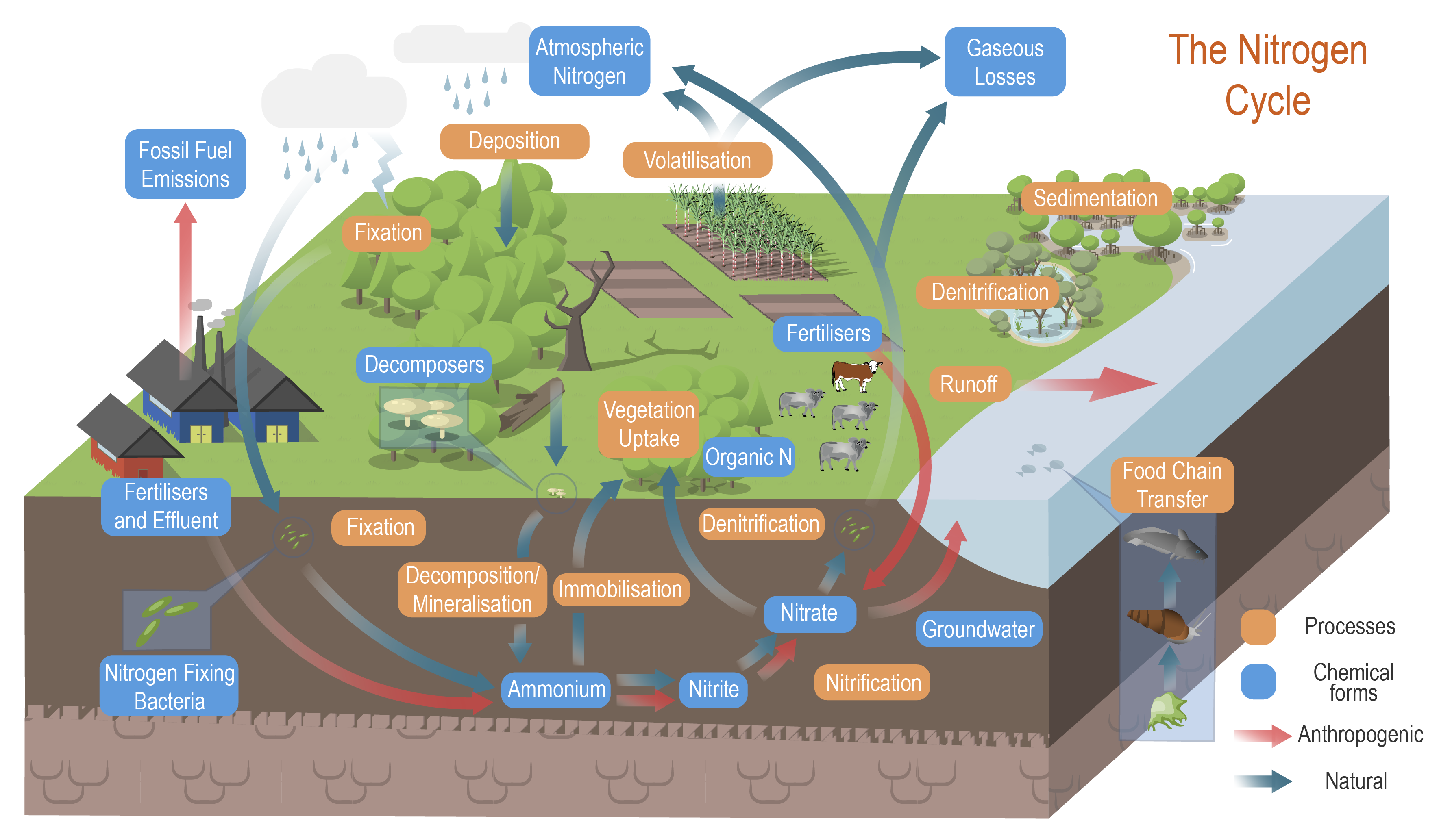
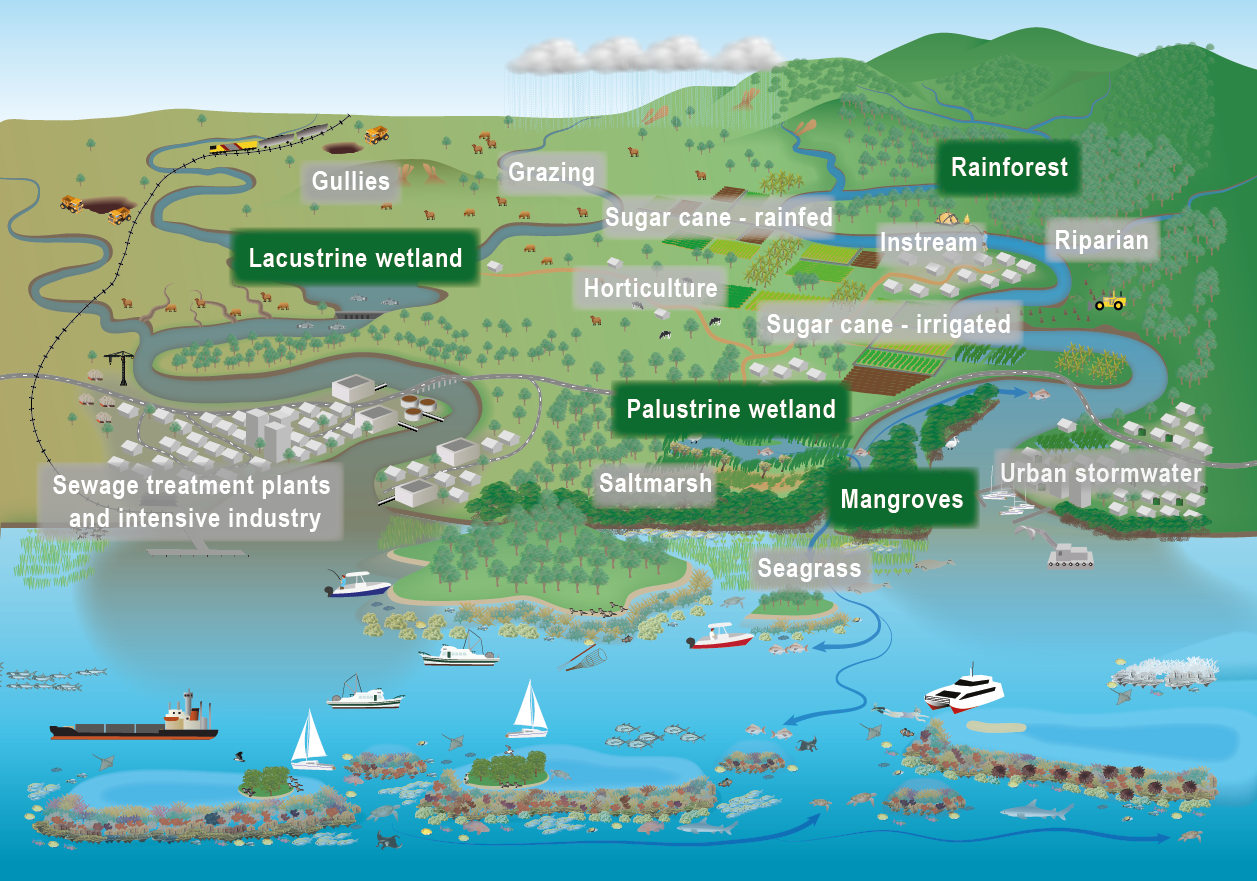
The conceptual models were compiled by researchers in collaboration with a wide range of stakeholders from Natural Resource Management groups, universities and government agencies and based on available scientific information[1]. Click on elements of the model or select from the tabs below The nitrogen cycleThe nitrogen cycle, in which atmospheric nitrogen is converted into different nitrogenous compounds, is one the most crucial natural processes to sustain living organisms. During the cycle, certain bacteria ‘fix’ atmospheric nitrogen into ammonia, which plants need in order to grow. Other bacteria convert the ammonia into amino acids and proteins. Animals eat the plants and consume the protein. Nitrogen compounds return to the soil through animal and plant waste. Bacteria convert the waste nitrogen back to nitrogen gas, which returns to the atmosphere. In an effort to make crops grow faster, people use nitrogen in fertilisers. However, excessive use of those fertilisers, as well as urban stormwater and wastewater discharges, can adversely affect the environment and human health, as it can contribute to the pollution of groundwater and surface waters. Nitrogen processes in the landscapeExcess nitrogen (N) is one of the most prevalent and serious pollutants affecting waterways. Agricultural, urban and industrial activities have increased the amount of reactive N globally by a factor of nine from pre-industrial times[2][5][1]. The N generated from land use and erosion in catchments may be transported by surface water runoff and groundwater flows to waterways and coastal and marine ecosystems, causing eutrophication and the degradation of these ecosystems[3]. As manufactured nitrogen production exceeds what is cycled naturally, the excess nitrogen in its different forms can harm natural ecosystems[7]. The amount and type of nitrogen and its effects, vary across ecosystems as it travels from its source to the sea[6]. Nitrogen is transported in water and can also be stored in sediments and plants, including algae. Algal blooms (both macroalgae and microalgae) and increased plant growth occur in areas with increased nutrients and high light availability. Different parts of the landscape process dissolved and particulate nitrogen differently depending on the soils, vegetation, hydrology, and sources of nitrogen and carbon. Some land units and land uses contribute nitrogen to waterways, others, like wetlands, remove it.
Additional information
Pages under this sectionReferences
Last updated: 28 September 2021 This page should be cited as: Department of Environment, Science and Innovation, Queensland (2021) Nitrogen processes and cycle, WetlandInfo website, accessed 8 May 2025. Available at: https://wetlandinfo.des.qld.gov.au/wetlands/ecology/processes-systems/nitrogen-concept-model/ |

 — Department of the Environment, Tourism, Science and Innovation
— Department of the Environment, Tourism, Science and Innovation